|
When lewis structure of BH 3 is observed, we can clearly see, there are no resonance structures for BH 3 because we cannot convert lone pairs to bonds or bonds to lone pairs with boron and hydrogen atoms. But, boron atom has not fulfilled its octal. There are no charges on boron and hydrogen atoms in borane. Are there charges on boron and hydrogen atoms in borane lewis structure BH3 This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. Boron atom gives 3 valence electrons and each hydrogen atom gives 1 valence electron. Science Chemistry Chemistry questions and answers Which of the following has a trigonal planar electron domain geometry Select one: a. The sketch we drawn above is the lewis structure of BH 3.Īsk your chemistry questions and find the answers BH 3 total valence electrons.So, there are no lone pairs to mark on boron and hydrogen atoms.Valence electrons pairs are remaining to draw the rest of the structure. There are three B-H bonds in the above sketch.From above two facts, we can decide boron should be the center atom of BH 3. 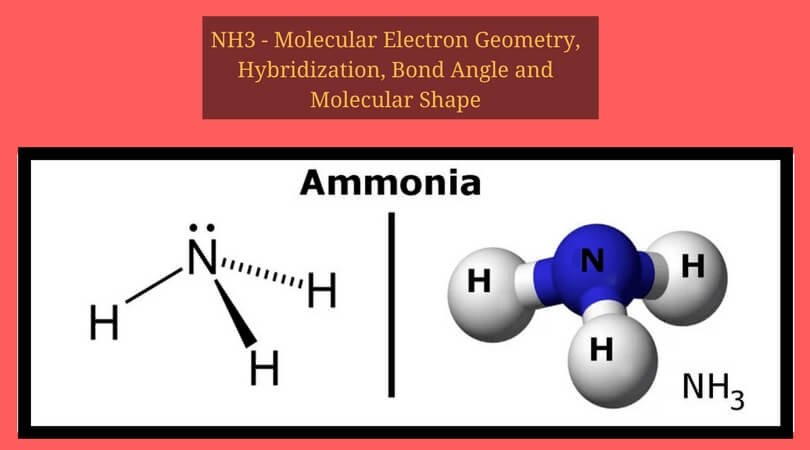
From that case too, boron has the highest chance to be the center atom. Therefore boron is moreĮlectropositive than hydrogen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
